The Risk Analysis Paradox
In regulated product development, risk analysis is one of the most formalized engineering activities. Hazard analyses are created, risk matrices are populated, and mitigation tables are carefully maintained.
Yet many design problems discovered during verification or even post-market could have been addressed much earlier.
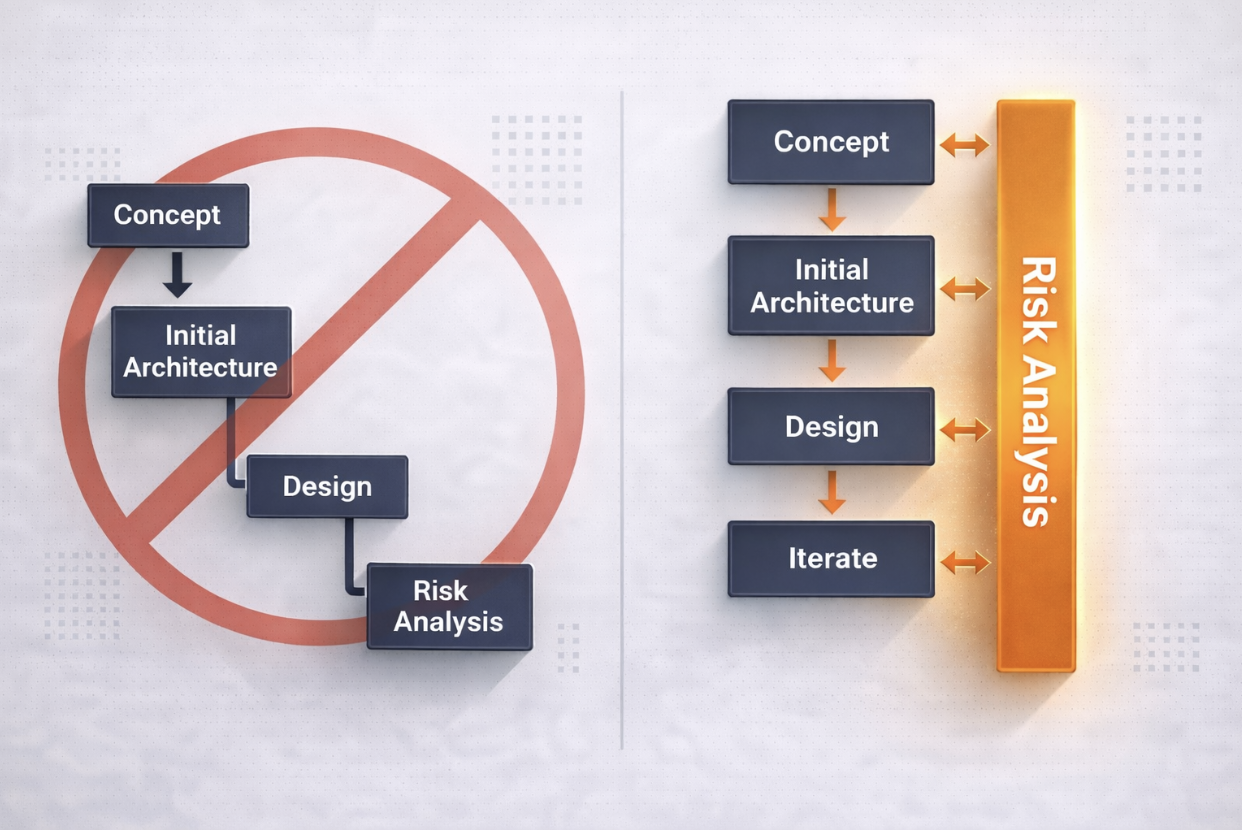
The issue is rarely the absence of risk analysis. Most programs perform it thoroughly. The more common problem is that risk analysis exists, but it does not meaningfully influence the design itself.
When this occurs, risk analysis becomes a documentation exercise. Hazards are recorded, mitigations are assigned, and traceability is maintained, but the design proceeds largely unchanged.
This outcome is typically structural. Risk analysis is introduced after key decisions are made and is not fully integrated into how engineering decisions are evaluated.
Risk Analysis Comes Too Late
The most consequential engineering decisions occur early in development. Product architecture, subsystem interfaces, energy transfer paths, and user interactions are typically defined during concept and early design phases.
In many programs, however, formal risk analysis begins once these decisions are already established. At that point, the activity naturally focuses on documenting hazards and identifying mitigations, but the opportunity to influence the design has already narrowed.
This timing shapes outcomes. When design flexibility is limited, teams tend to rely on labeling, instructions for use, or training rather than design changes. This does not reflect preference. It reflects that the architecture is already set.
For risk analysis to influence engineering decisions, it must be present earlier. This means engaging when alternative concepts are still viable and tradeoffs are still being made.
Integrating Risk into Early Design Decisions
Incorporating risk analysis earlier in development does not require a new process. It requires using existing engineering activities differently.
Three practices are consistently effective.
- Evaluate risk alongside architecture decisions
During concept development, teams routinely compare alternatives based on performance, cost, and feasibility. Risk should be evaluated at the same time.
For each architecture option, ask:
- What failure mechanisms are inherent to this concept?
- Which hazards are eliminated or introduced by this approach?
For example, two concepts may meet the same functional requirement but differ significantly in how energy is transferred or controlled. One may inherently limit exposure to hazardous states, while the other depends on tight tolerances or user behavior to remain safe.
When risk is evaluated at this stage, it becomes a selection criterion, not a downstream check.
- Frame risk in terms of failure mechanisms, not categories
Early risk discussions are most effective when grounded in how the system can fail physically or functionally.
Rather than listing abstract hazards, teams should describe how energy is transferred, where control can be lost, and how user interaction could create unintended states.
For example, describing “uncontrolled motion due to loss of braking torque” is far more actionable than “device malfunction.” The former immediately suggests design paths such as redundancy, passive braking, or fail-safe geometry.
This level of specificity connects risk directly to engineering decisions.
- Use risk reviews as design challenges, not status checks
Early risk reviews should not focus on completeness. They should focus on whether the design assumptions are valid.
Effective discussions center on questions such as:
- What would have to be true for this design to fail?
- Where are we relying on ideal conditions or perfect user behavior?
- What design change would remove this hazard entirely?
In practice, this often reveals that a hazard persists not because it is unavoidable, but because a particular design direction has not been questioned.
When risk reviews are structured this way, they become a mechanism for surfacing better design alternatives, not just documenting current ones.
Risk Analysis Should Change Engineering Decisions
Risk analysis is not valuable because it produces a complete hazard table. It is valuable because it influences engineering decisions.
When risk activities begin early and are embedded in how design decisions are evaluated, they become one of the most effective tools for improving product safety and robustness.
If risk analysis does not influence engineering decisions, it is documentation, not risk management.
Free Risk Integration Assessment
If your risk analyses are not leading to design changes, the issue is rarely the quality of the analysis itself. More often, it is how and when risk is integrated into development.
A65 works with leadership teams to evaluate how risk management is applied within product development. A focused assessment often reveals where risk is introduced too late, where it operates outside engineering decision-making, and where opportunities to improve the design are missed.
These gaps are not typically visible within the program. However, they often explain why known risks persist into verification and beyond.
If you would like an objective assessment of how risk is influencing your design decisions, we welcome the conversation.
Email: sdonnigan@a65consulting.com
Or schedule your review online
References
ISO 14971:2019. Medical devices — Application of risk management to medical devices. International Organization for Standardization.
ISO 13485:2016. Medical devices — Quality management systems — Requirements for regulatory purposes. International Organization for Standardization.
IEC 60601-1:2005+AMD1:2012+AMD2:2020. Medical electrical equipment — General requirements for basic safety and essential performance. International Electrotechnical Commission.
AAMI TIR57:2016. Principles for medical device security—Risk management. Association for the Advancement of Medical Instrumentation.
INCOSE. (2015). Systems Engineering Handbook: A Guide for System Life Cycle Processes and Activities (4th ed.). Wiley.
U.S. Department of Defense. (2012). Systems Engineering Guide for Systems of Systems.