by Steve Donnigan | May 1, 2026 | Uncategorized
Verification failures are rarely surprises. When a test fails late in development, teams often treat it as an execution issue. Attention shifts to the method, the setup, or the result itself. In most cases, the underlying problem was introduced much earlier. Many...

by Steve Donnigan | Apr 24, 2026 | Medical Device
Outsourcing is often presented as a straightforward way to move faster. If a program needs more speed, add external resources. If specialized expertise is missing, bring in a partner. If timelines are tight, increase capacity. Sometimes this works exactly as intended....

by Steve Donnigan | Apr 17, 2026 | Uncategorized
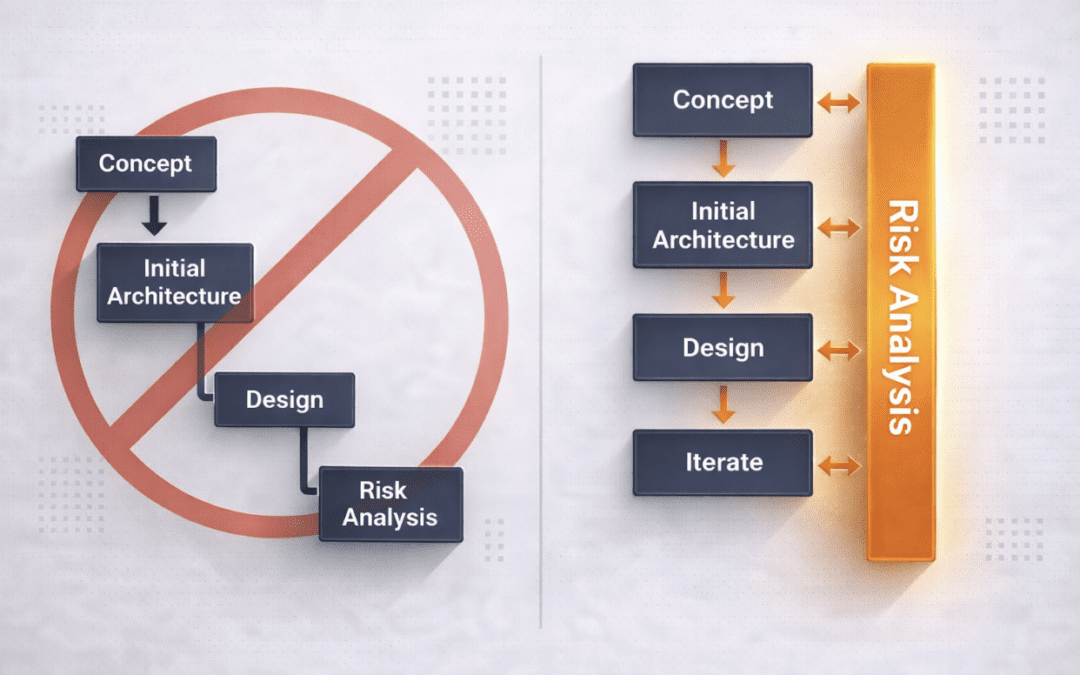
The Risk Analysis Paradox In regulated product development, risk analysis is one of the most formalized engineering activities. Hazard analyses are created, risk matrices are populated, and mitigation tables are carefully maintained. Yet many design problems...

by Steve Donnigan | Apr 6, 2026 | Uncategorized
Strong engineering teams still miss milestones. Many medical device companies have excellent engineers, experienced quality groups, and significant investment in development programs. Yet milestones still slip. This pattern appears across organizations of every size....

by Steve Donnigan | Mar 19, 2026 | Uncategorized
Medical device development is a coordinated system of distinct domains operating under regulatory constraint. When that system performs well, progress feels disciplined and predictable. When it struggles, the issue is often not capability but structure. Sustained...

by Steve Donnigan | Mar 11, 2026 | Uncategorized
Most development teams believe they understand their users. Human factors activities are completed, documented, and validated. Yet use-related problems still emerge once devices enter real clinical environments. These failures are rarely random. They typically stem...

by Steve Donnigan | Mar 5, 2026 | Uncategorized
Believing in DfM Is Not the Same as Doing It Most development leaders agree that Design for Manufacturing belongs in early product development. The logic is clear. Cost, quality, and scalability are shaped long before design transfer. Yet many organizations that...

by Steve Donnigan | Feb 25, 2026 | Uncategorized
Cost, Quality, and Predictability Design for Manufacturing is often discussed as a downstream optimization activity that is addressed after the design is complete and the focus shifts to scale-up and production. In medical device development, that framing is...

by Steve Donnigan | Feb 12, 2026 | Uncategorized
Designing a medical device that performs beautifully isn’t enough. It also has to meet the world’s toughest regulatory and safety standards. In 2026, compliance is more than just a checkbox, but, rather, a competitive advantage. Teams that integrate regulation...

by Steve Donnigan | Feb 6, 2026 | Uncategorized
Cutting prototyping to save time or expense is one of the most reliable ways to lengthen a medical device development program. When prototyping is undervalued and treated as an optional expense rather than a requirement, cost and schedule risk almost always increase...