by Steve Donnigan | Feb 25, 2026 | Uncategorized
Cost, Quality, and Predictability Design for Manufacturing is often discussed as a downstream optimization activity that is addressed after the design is complete and the focus shifts to scale-up and production. In medical device development, that framing is...

by Steve Donnigan | Feb 12, 2026 | Uncategorized
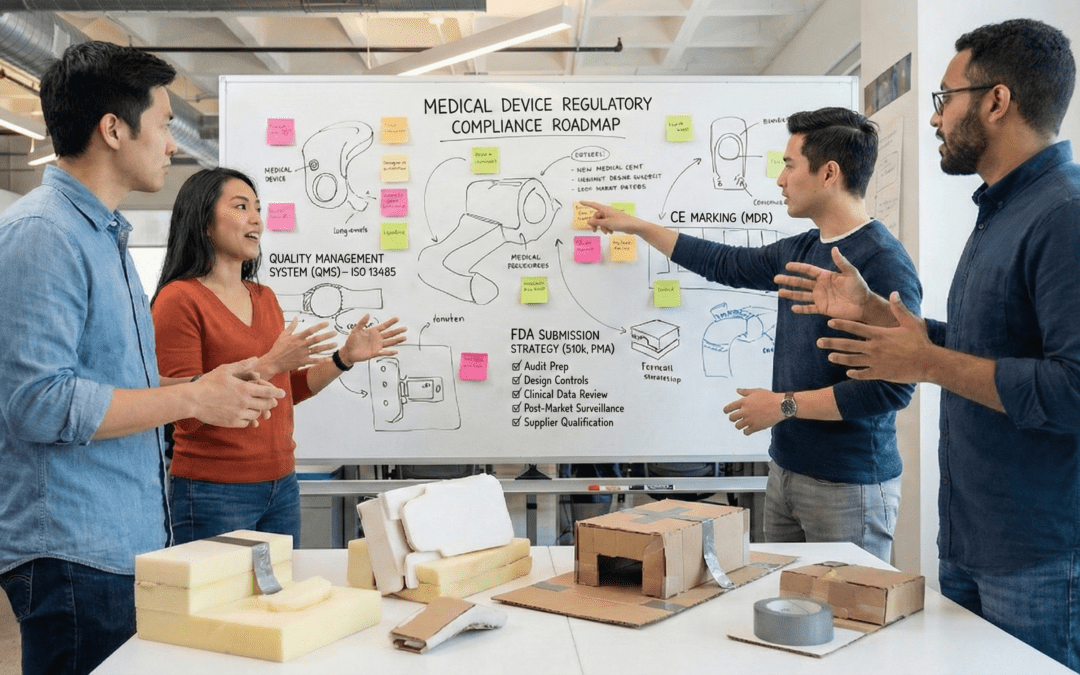
Designing a medical device that performs beautifully isn’t enough. It also has to meet the world’s toughest regulatory and safety standards. In 2026, compliance is more than just a checkbox, but, rather, a competitive advantage. Teams that integrate regulation...

by Steve Donnigan | Feb 6, 2026 | Uncategorized
Cutting prototyping to save time or expense is one of the most reliable ways to lengthen a medical device development program. When prototyping is undervalued and treated as an optional expense rather than a requirement, cost and schedule risk almost always increase...

by Steve Donnigan | Jan 28, 2026 | Uncategorized
Medical device organizations today are operating in a tighter, more complex environment than ever before. Regulatory scrutiny continues to rise, technology stacks are converging, development timelines are compressing, and cost pressure is unrelenting. In this context,...

by Steve Donnigan | Jan 21, 2026 | Uncategorized
Outsourcing Engineering Is a Strategic Business Decision Medical device startups rarely fail because of insufficient engineering effort, they fail because scarce resources such as capital, time, and attention are misallocated. Outsourcing engineering is often...

by Steve Donnigan | Dec 12, 2025 | Uncategorized
How to Get Them Right Designing a successful medical device is a disciplined process rooted in systemized product development and advanced regulatory compliance, not luck or guesswork. Every life saving product on the market today has passed through FDA-mandated...