MEDICAL DEVICE DESIGN AND ENGINEERING
Facing a challenging project that requires additional engineers or expertise?
seamlessly integrate with your team and work alongside them to deliver amazing results!
Mechanical Engineering
Need an end-to-end mechanical engineering services solution?
We will brainstorm solutions, generate multiple concepts, build mathematical models if required, complete detailed designs, and build and test prototypes. Our team can also help with design changes for cost reduction purposes. Trust us to deliver efficient and effective solutions for your product development challenges.
Mechanical Engineering Services:
- Precision mechanism design
- High volume disposable device design
- Hardware and electromechanical system design
- Complex robotic and automated system design
- Electronic enclosure design
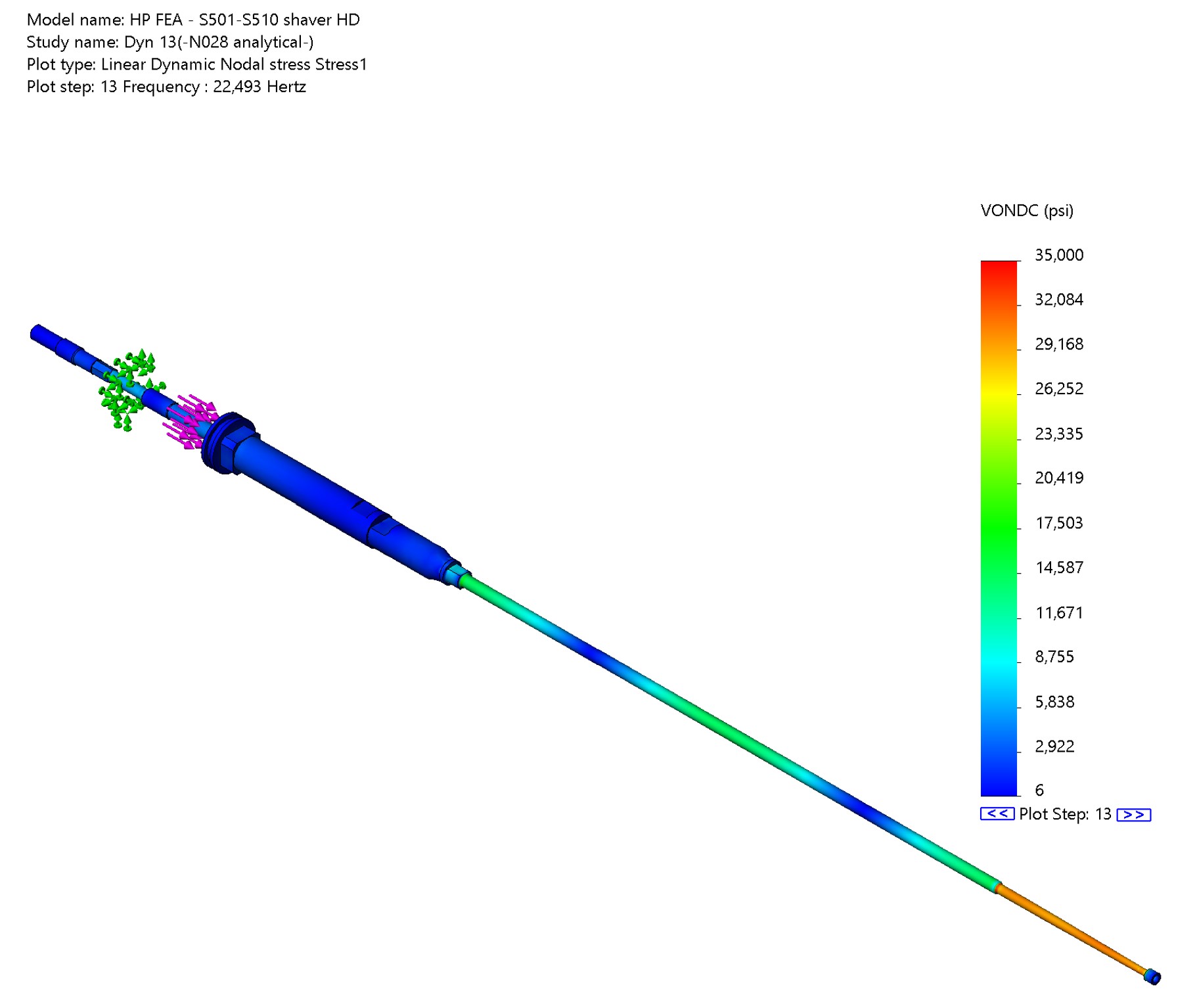
- FEA and classical engineering analysis
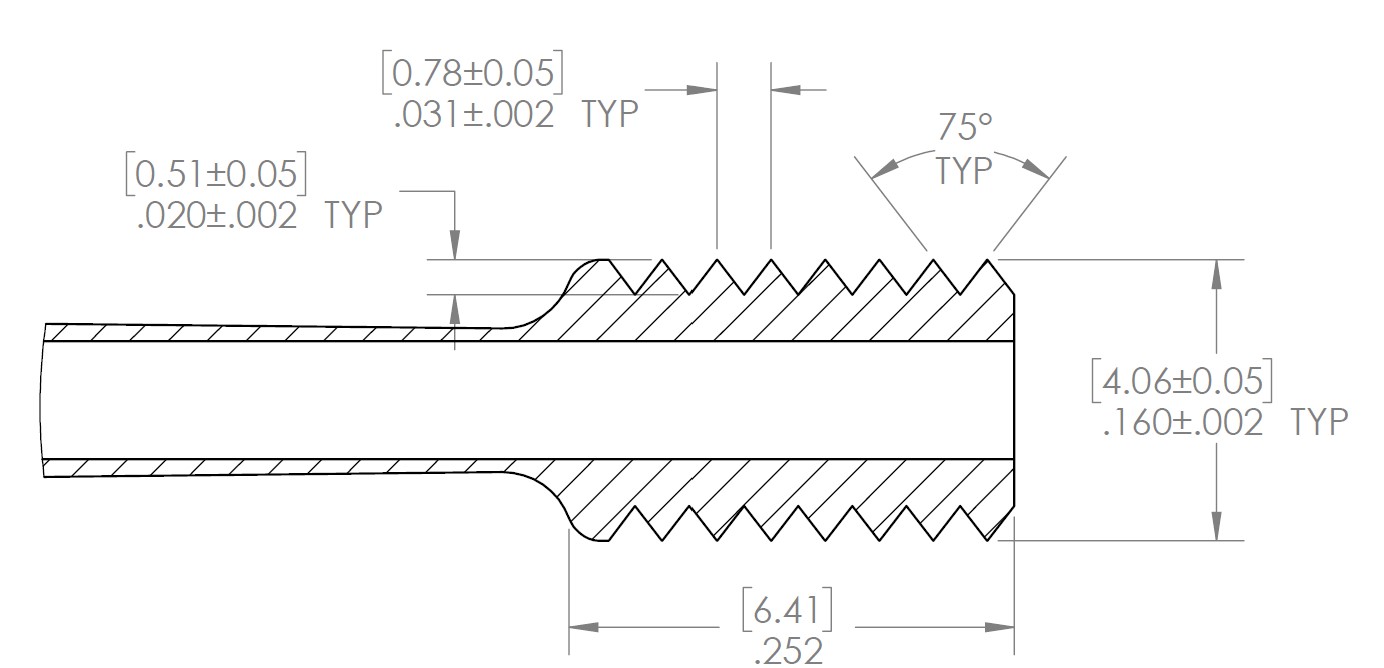
- Tolerance analysis with GD&T
- Thermal design
- SolidWorks CAD modeling and drawing creation
System Engineering
We can develop a system architecture that meets the product requirements and project objectives, and implement risk mitigations derived from hazard analysis and FMEA. Our expertise includes traceability, verification and validation protocols and reports, human factors engineering, VOC surveys and interviews, and system troubleshooting with root cause investigation.
We focus on delivering efficient solutions that ensure your product is safe, user-friendly, and meets all requirements.
Manufacturing Engineering
We implement design for manufacturing/assembly (DFM/DFA) principles to optimize the production process and reduce costs, enabling you to maximize your profits. Our services include COGS estimation, fixture and tooling development, investigating and solving production problems, manufacturing instruction creation, production line layout and balancing, supplier selection and management, and efficiency improvements.
You can count on us to deliver efficient and effective solutions for your manufacturing engineering needs.
Program Management
Need support managing complex programs across multiple engineering initiatives?
A65 Consulting provides comprehensive program management services to help you coordinate multiple concurrent projects with clarity and control. We go beyond traditional project management by delivering full visibility into timelines, resources, and risks across your entire engineering portfolio. Our team partners with your organization to keep programs aligned, on track, and moving forward without adding unnecessary overhead.
Program Management Services:
- Program-level roadmap development and multi-project schedule coordination
- Gantt chart creation and ongoing schedule management
- Cross-functional task ownership and accountability tracking
- Milestone tracking to ensure on-time, in-scope delivery
- Resource planning and gap identification across teams and budgets
- Proactive risk identification and mitigation planning
- Executive-level status reporting and stakeholder communication
- Coordination of multiple projects in the absence of a formal PMO or to supplement your PMO

Quality Engineering
Need expert guidance to ensure your medical device meets rigorous quality and regulatory standards?
A65 Consulting provides experienced quality engineering support to help medical device organizations establish and maintain compliant quality systems. Working collaboratively with all cross functional groups throughout the product life cycle, our team integrates seamlessly with client organizations to implement practical, FDA-aligned solutions. With decades of embedded industry experience, we deliver the structure, oversight, and documentation required to support successful product development and regulatory readiness.
Quality Engineering Services:
- Quality Management System (QMS) design and implementation, including FDA-required procedures and documentation
- Supplier audits to ensure regulatory compliance across the supply chain
- Internal audits that simulate regulatory authority audits and assess organizational readiness
- CAPA (Corrective and Preventive Action) and nonconformance management support
- Preparation and maintenance of quality documentation for regulatory submissions and regulatory compliance
- Ongoing quality engineering support for new and existing product development programs
- Fractional and embedded quality engineering services tailored to organizational needs

Regulatory Affairs / Support
Need guidance in navigating the regulatory pathway for your medical device?
A65 Consulting provides regulatory affairs support focused on the preparation, documentation, and technical rigor required to meet global regulatory requirements and maintain FDA/OUS regulatory compliance. We work behind the scenes to ensure submissions are accurate, thorough, and well-supported while your internal regulatory team maintains ownership of all direct communications with the regulatory authorities. Our approach increases your regulatory bandwidth without disrupting established processes or leadership.
Regulatory Affairs & Support Services:
- Global regulatory submission support, including technical documentation, data summaries, and supporting evidence
- Review of device designs and development documentation for compliance with applicable regulatory standards
- Preparation of background regulatory materials for regulatory submissions
- Regulatory pathway analysis and strategic guidance for device classification and clearance
- Support for responses to regulatory body questions and additional information requests
- Coordination with cross functional teams to ensure regulatory alignment throughout product development
- Documentation support to maintain ongoing compliance to global regulatory requirements and audit readiness

